ATTENTION: ASSAY MANUFACTURERS
Grow your assay sales! Enhance your LFAs with new capabilities.
Pair your assays with the Assaya Intelligent Analyzer universal reader to deliver machine-verified, quantitative results that move beyond positive/negative with results securely stored in the cloud. Expanding markets, improving real-world performance, and increasing customer confidence, sales and adoption.
WHY PARTNER WITH ASSAYA?
Turn your assays into smarter, quantitative connected solutions
When your lateral flow assays are certified for use with the Assaya iaX Intelligent Analyzer they become more valuable and scalable than standalone rapid tests. Machine-read interpretation, quantitative outputs and instant records, so buyers say “yes” faster.
When certified for use with iaX, your LFAs become precise, quantitative, and cloud-ready — eliminating buyer objections and accelerating sales and adoption.
Elevate LFA beyond simple positive/negative. iaX provides a precise Quantifiable Unit (QU) result. Test lines become a precise, reproducible number — no subjective calls.
Computer vision reduces human read errors. Machine-read interpretation delivers consistent, objective results across operators and sites.
LFA machine-read results are encrypted, timestamped, and securely stored in the cloud. Integrates with customer systems such as EHR/LIMS for auditable real-time reporting.
Unlock clinics, pharmacy, EMS, field, farm, and industrial markets without building your own reader. iaX certifications span US FDA, IVD-CE, IVDR-EU, ANVISA, and more.
No need to delay or outlay to build your own hardware. Leverage iaX instead of building and validating a proprietary reader stack.
COMPATIBILITY & QUALITY
iaX-Certified Badge & qualification
After your assays are profiled, and verified to read correctly on iaX devices, we provide digital assets that you can use to promote your iaX certification:
- On your website
- Product Packaging
- In your IFU
We will also designate your assays as iaX-qualified in the Assayverse, so they can be found more easily by prospective customers.

The iaX-Qualified Badge
- Assay profile (geometry, timing, thresholds) mapped to iaX
- QC/Invalid detection behaviors defined
- Calibration curve (dynamic range; LoD/LoQ mapping) recorded
- Change-control & versioning registered (lot/config files)
- Catalog listing with use-case notes and data outputs
FROM DISCOVERY TO LISTING
Get your assays iaX-qualified in weeks, not quarters
Qualification and certification can be turned around in short order, subject to availability of your assays and controls. Certification is available in Europe, Taiwan, China, and USA.
Use-cases, matrices, targets, and timeline defined collaboratively with your team
Cassette geometry, timing windows, controls, and read behavior mapped on the iaX reader
Thresholds, curve fit, and invalid rules established and validated against your reference data
IFU language, reader prompts, and training assets prepared for your review and approval
Badge issued, assay published to the iaX library, and co-marketing begins. Typical pilot: 2–6 weeks.
Get the iaX Certification Process documentation and outline emailed to your inbox immediately.
EXACTLY WHAT WE NEED
Inputs we’ll ask from your team
Clear, lightweight checklist to keep momentum. Most manufacturers can provide these within days.
- Cassette & line layout: geometry drawing (DXF/SVG/PDF)
- IFU read timing: min/target/max; revisit drift/over-read risks
- Sample matrices & volume: blood/serum/saliva/urine; water/feed/leaf, etc.
- Controls & behavior: control/test lines, invalid states, hook effect notes
- Performance data: LoD/LoQ, dynamic range, cross-reactivity highlights
- Environmental tolerances: temp/lighting ranges for field use
- Labelling/ID: lot/expiry format; optional QR/2D codes for metadata
- Data model fields: analyte, unit, range, reference thresholds
BENEFITS FOR TEST KIT DEVELOPERS
Turn your LFA into a connected, quantitative, machine-read diagnostics solution
Reduce variability, add quantitative outputs, and deliver audit-ready cloud records — unlocking new markets and faster adoption paired with the iaX Intelligent Analyzer.
- Access to new buyers & use-cases Unlock EMS, pharmacy, clinic, field, farm, and industrial markets without building your own reader.
- Faster purchase cycles Machine-read, cloud-ready tests remove buyer objections around variability and record-keeping.
- Bigger deal sizes Bundle tests with the iaX platform (hardware + software) for higher average order value.
- Tender eligibility “iaX-Qualified” status helps meet machine-read/traceability requirements in RFPs.
- Consistent interpretation Computer-vision reading reduces operator variability vs human interpreted visual reads.
- Quantitative outputs Elevate qualitative strips with numbers clinicians can act on and trend over time.
- Built-in QC & invalid detection Automated checks prompt retests and cut false calls/returns
- Better real-world performance Standardised optics and read windows improve reproducibility across sites
- Instant cloud records (AssayaDX) Results auto-sync for dashboards, cohort tracking, and remote review
- APIs & exports Easy movement of data to LIMS/EHR/ERP for enterprise adoption
- Post-market evidence Aggregated (de-identified) performance data supports publications and label expansions.
- Usage analytics Understand adoption by site, assay, and indication to guide inventory and roadmap.
- Rapid qualification path Assay profiling, timing windows, and threshold mapping to get you live quickly
- No hardware investment Leverage iaX instead of building/validating your own proprietary reader stack
- SDK & tech support Engineering guidance, test plans, and validation templates to streamline onboarding.
- Backwards-compatible updates Firmware/algorithm releases maintain assay support without breaking changes
- Compatibility listing & badge “iaX-Qualified” placement in Assaya’s catalog and marketing materials.
- Global reach Tap into Assaya’s network across healthcare, agriculture, aquaculture, food safety, and environment.
COMPATIBILITY & QUALITY
Frequently asked questions regarding iaX Certification for Lateral Flow Assays
Your assay is profiled, and verified to read correctly on iaX across defined lots and conditions.
General / Assay Compatibility
+What does it mean for an LFA to be “iaX-Qualified”?
+Which LFAs can the iaX read?
+Do you support multiplex strips?
+Can iaX provide quantitative results?
+What happens if we change cassette geometry or chemistry?
Timing, QC & Configuration
+What are “timing windows,” and why do they matter?
+How does iaX handle invalid tests or weak control lines?
+Can we set custom thresholds or reflex logic?
+How do you manage lot-to-lot variation?
+What environmental ranges are supported (temp/light)?
Data & Integration
+Where do results go?
+Can we export or integrate with our systems?
+Who owns the data?
+What about offline sites?
+Can we add custom fields to the result payload?
Hardware & Operator Experience
+How big is the iaX? Is it field-ready?
+What does the operator experience look like?
+Can we brand the UI or reports?
+How many tests can be run per device per day?
Regulatory & Compliance
+What is iaX’s regulatory status?
+Will this help with audits or tenders?
+How are firmware/algorithm updates handled?
+Can iaX help us tighten our IFU?
Commercial & Getting Started
+What’s the benefit of becoming iaX-Qualified?
+Do you offer co-marketing or channel support?
+Can we sell our assays bundled with iaX?
+What does the Developer Kit include?
+How long does qualification take?
+What does it cost?
GET YOUR ASSAYS QUALIFIED
Start the conversation
Tell us about your lateral flow assays and target markets. Our qualification team will follow up with the process details and timeline.
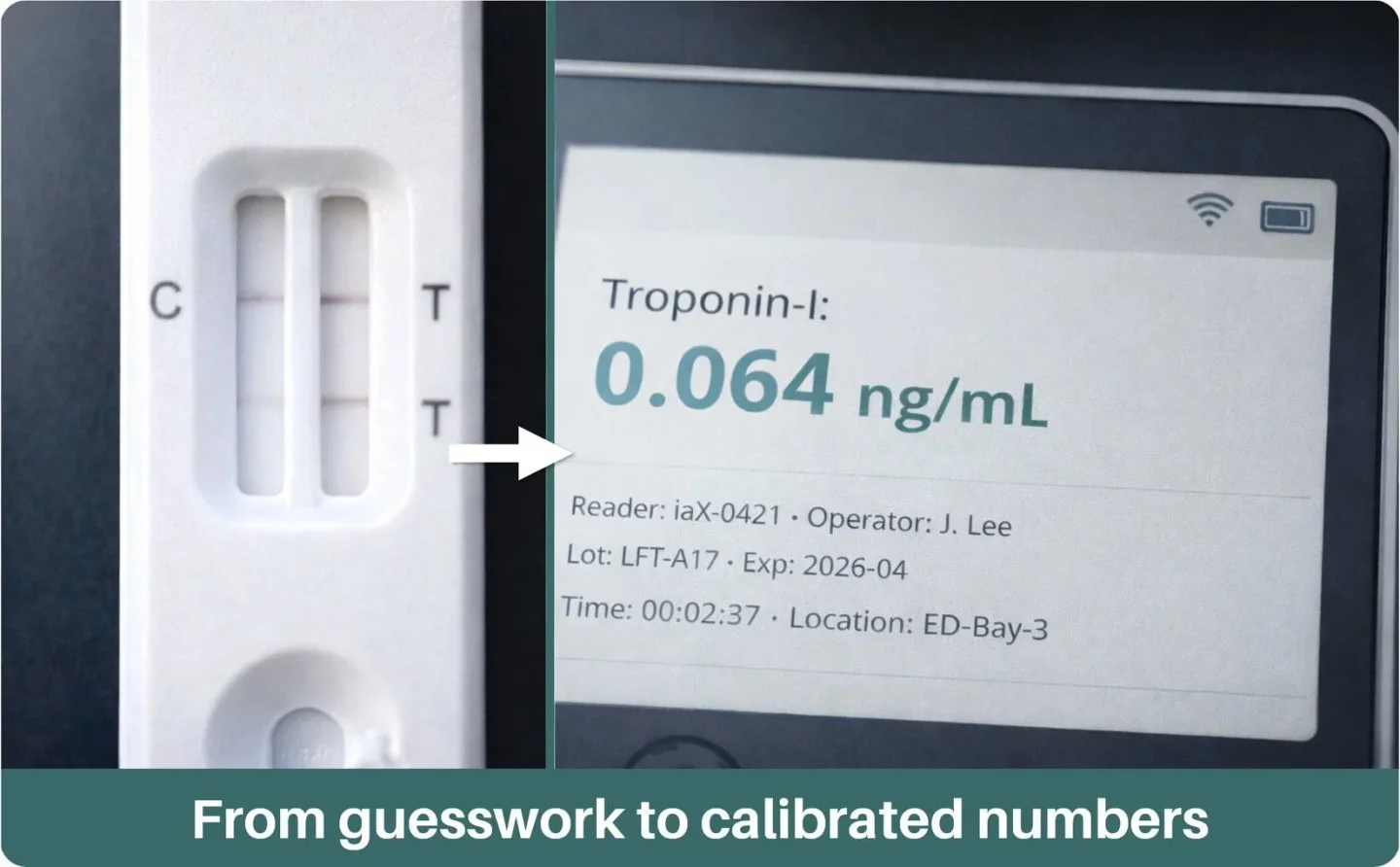
Get your lateral flow assays iaX-qualified for machine-read, quantitative, cloud-connected results — typically in 2–6 weeks. Unlock new markets and accelerate adoption with the iaX Intelligent Analyzer.



